Boston, Mass., May 6, 2015 – The Institute for Clinical and Economic Review (ICER) has released a new Evidence Report titled Treatment Options for Relapsed or Refractory Multiple Myeloma: Effectiveness, Value, and Value-Based Price Benchmarks. The report, which incorporates revisions based on feedback from stakeholders received during a formal public comment period, provides analyses of the clinical effectiveness, long-term cost-effectiveness, and potential budget impact of treatments for multiple myeloma. In addition, the results of these analyses are used to calculate value-based price benchmarks for the drugs. These price benchmarks reflect the drugs’ long-term cost-effectiveness for patients while also noting the price at which their potential short-term budget impact raises serious concerns for affordability across the entire health care system.
ICER’s report focuses on adults with multiple myeloma whose disease has not responded to at least one previous line of treatment (i.e., refractory) or has relapsed following such treatment, are not currently on maintenance treatment, and are not being considered for stem cell transplant. The report will be the subject of the May 26, 2016 inaugural meeting of the Midwest Comparative Effectiveness Public Advisory Council (Midwest CEPAC) in St. Louis, MO.
A draft version of the report was open to a two-week public comment period. During this period, ICER received 14 sets of public comments from manufacturers, clinicians, and patients. We then made changes to the report based on this valuable input. ICER encourages all stakeholders to be engaged in our process to ensure that we deliver a comprehensive and actionable report.
Our review focused on the following treatment regimens for relapsed and/or refractory multiple myeloma, including second-line and/or third- or later-line use:
- Carfilzomib (Kyprolis®) with lenalidomide (Revlimid®) and dexamethasone (CFZ+LEN+DEX)
- Ixazomib (Ninlaro®) with LEN+DEX (IX+LEN+DEX)
- Elotuzumab (Empliciti™) with LEN+DEX (ELO+LEN+DEX)
- Panobinostat (Farydak®) with bortezomib (Velcade®) and DEX (PAN+BOR+DEX)
- Pomalidomide (Pomalyst®) with low-dose DEX (POM+LoDEX)
- Daratumumab (Darzalex™) monotherapy (DARA)
In available clinical studies, these regimens were compared to LEN+DEX, BOR+DEX, or high-dose DEX (see summary of report findings for specific comparators), except for DARA, which did not have a comparator in the clinical trials available during our review.
A key goal of ICER’s reports is to ensure that patients, providers, insurers, and policymakers have the information they need to support efforts to improve the quality and value of care. Highlights of the report’s findings include:
Comparative Clinical Effectiveness: There are no head-to-head studies of the newer drug regimens for multiple myeloma, and limitations in the strength of evidence on these treatments allow only moderate certainty in their overall net health benefit compared to earlier standards of care. Nonetheless, for most of the newer drugs, we judged the evidence adequate to demonstrate at least a small net improvement, based primarily on accepting increases in “progression-free survival” as a meaningful patient-centered outcome. For some drugs, however, we judged the level of clinical benefit to be less certain or the evidence to be insufficient for at least one type of use (second- or third- or later-line use). Further detail is provided in the Appendix to this press release below.
Long-term Cost-Effectiveness: We developed a computer model to simulate the clinical outcomes and costs of different treatment regimens in order to compare their long-term cost-effectiveness. This model required assumptions linking improvement of progression-free survival to overall survival, a correlation that some experts feel is inconsistent at best. Results of our model showed that for use as second-line therapy, each of the treatment regimens results in a gain of approximately one year of overall survival relative to the standard of care against which these treatments were studied to gain FDA approval. Treatments increased length of life but at a significant added cost. The incremental costs ranged from a low of approximately $173,000 for CFZ+LEN+DEX to approximately $354,000 for ELO+LEN+DEX versus LEN+DEX. Cost-effectiveness vs. LEN+DEX was estimated to be $200,000 per quality-adjusted life year (QALY) gained for CFZ+LEN+DEX, $428,000 for ELO+LEN+DEX, and $434,000 for IX+LEN+DEX. While these results varied, all exceeded commonly-cited thresholds of $50,000 to $150,000 per quality-adjusted life year (QALY).
Cost-effectiveness results for the above regimens were similar for third- or later-line use. In comparison to LEN+DEX, however, the use of PAN+BOR+DEX was found to be both more effective and less expensive. These results should be interpreted with caution, however, as the clinical data supporting PAN+BOR+DEX have been called into question (see Appendix). Neither DARA nor POM+LoDEX were included in the model because these regimens were studied in patients with more advanced disease.
Potential Budget Impact: Given the relatively small patient population for these treatments, our estimate of the potential aggregate costs of treatment with new regimens does not reach the level at which we estimate it would raise a notable concern for affordability across the entire health system.
Value-based Price Benchmarks: Achieving levels of value more closely aligned with patient benefit would require substantial discounts from the list price in many cases, and in other cases, there is no realistic price for the newest agents that would achieve these thresholds. This phenomenon of requiring discounts approaching 100% to reach standard cost-effectiveness levels is well known and relates to situations in which current treatment is already near or beyond the cost-effectiveness threshold. Adding even more expense with a new treatment on top of existing treatment, as is the case for multiple myeloma drugs, means that to reach standard cost-effectiveness levels the entire regimen, including the older, existing drugs that are part of the regimen, would need to be deeply discounted. Specific dollar values can be seen in the table below, and additional material is provided in the report.
“The introduction of newer regimens for second- and third-line use in multiple myeloma appears to confer clinical benefits in terms of lengthening progression-free survival, as well as improved quality of life. However, at current wholesale acquisition costs, the estimated long-term cost-effectiveness of these regimens exceeds commonly-cited thresholds,” noted ICER’s President Steven D. Pearson, MD, MSc.
The report, along with a revised set of voting questions, all public comments received, a summary of public comments, and ICER’s response to comments, are available on the ICER website.
The Evidence Report will be the subject of deliberation and vote at the inaugural meeting of the Midwest Comparative Effectiveness Public Advisory Council (Midwest CEPAC) on May 26, 2016 in St. Louis, MO. During that meeting, Midwest CEPAC members will publicly deliberate and vote on the evidence as presented in the report and discuss the findings with a policy roundtable of experts to discuss implications of the findings for policy and practice. The meeting will be free and open to the public. There will be a limited time during the meeting for any interested stakeholders to make an oral public comment on the report. Requests to make an oral comment should be submitted to publiccomments@icer-review.org no later than Thursday, May 19, 2016 at 5PM CT.
About ICER
The Institute for Clinical and Economic Review (ICER) is an independent non-profit research institute that produces reports analyzing the evidence on the effectiveness and value of drugs and other medical services. ICER’s reports include evidence-based calculations of prices for new drugs that accurately reflect the degree of improvement expected in long-term patient outcomes, while also highlighting price levels that might contribute to unaffordable short-term cost growth for the overall health care system.
ICER’s reports incorporate extensive input from all stakeholders and are the subject of public hearings through three core programs: the California Technology Assessment Forum (CTAF), the Midwest Comparative Effectiveness Public Advisory Council (Midwest CEPAC) and the New England Comparative Effectiveness Public Advisory Council (New England CEPAC). These independent panels review ICER’s reports at public meetings to deliberate on the evidence and develop recommendations for how patients, clinicians, insurers, and policymakers can improve the quality and value of health care. For more information about ICER, please visit ICER’s website.
Report Highlights
ICER Evidence Ratings
We judge there to be moderate certainty that CFZ, ELO, and IX, in combination with LEN+DEX, provide an incremental or better net health benefit for both second-line and third-line or subsequent therapy in adult patients with relapsed/refractory multiple myeloma relative to LEN+DEX alone. There is moderate certainty because while only one Phase III study was available for each regimen, the studies of focus had large patient populations and were of higher quality. Data on side effects do not demonstrate a systematic overall advantage for any of these regimens. We therefore assign the current body of evidence on the comparative clinical effectiveness of CFZ, ELO, and IX a “B+” rating using the ICER Evidence Rating Matrix.
PAN was rated as “promising but inconclusive” (“P/I”) for third- or later-line use. While study findings suggest clinical improvement over BOR+DEX, particularly in the subset of patients who had tried specific prior therapies, there is greater uncertainty in this evidence due to high rates of severe adverse events and study discontinuation in clinical trials. This rating was also given to POM, as the clinical benefit seen was in comparison to a regimen (high-dose DEX) that is not frequently used in clinical practice.
Evidence was determined to be insufficient (“I”) to determine a net health benefit for the remaining therapies and/or lines of use.
ICER Evidence Ratings, by Regimen and Line of Therapy
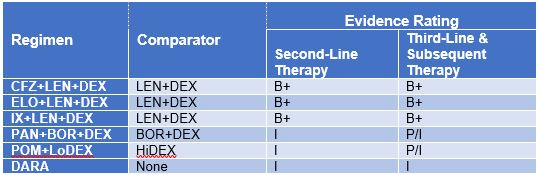
B+=Incremental or Better, P/I = Promising but Inconclusive, I = Insufficient
Comparative Value
The primary aim of our analysis of incremental costs per outcomes achieved was to estimate the cost-effectiveness of various treatments for patients with MM who have received one or two previous therapies (i.e., second- or third-line treatment) relative to LEN+DEX treatment. The model analyzed second- and third-line treatments separately. Note that we were unable to include DARA or POM+LoDEX in our analyses, as patients in these trials had more advanced disease than those receiving the other regimens of interest.
Use of each of the second-line regimens resulted in a gain of approximately one year of survival relative to LEN+DEX, which was split relatively evenly between the pre-progression and progressed health states.
Incremental costs ranged from a low of approximately $173,000 for CFZ+LEN+DEX to approximately $354,000 for ELO+LEN+DEX versus LEN+DEX, nearly all of which were driven by increased drug costs rather than progression, supportive care, or adverse event costs. Cost-effectiveness vs. LEN+DEX was estimated to be $200,000 per quality-adjusted life year (QALY) gained for CFZ+LEN+DEX, $428,000 for ELO+LEN+DEX, and $434,000 for IX+LEN+DEX.
Clinical and Economic Outcomes in the Second-Line
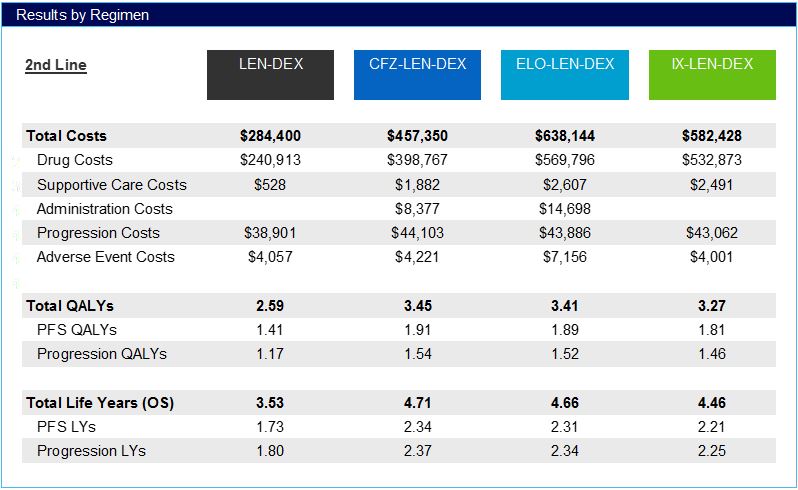
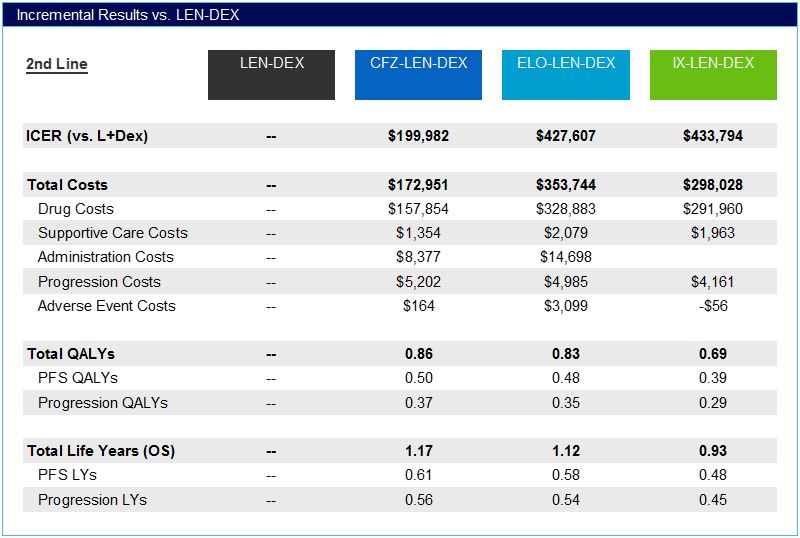
Table for third- line treatments is available in the report on page 50.
Potential Budgetary Impact and Value-Based Price Benchmark
The draft value-based price benchmark results are shown in the table below for 2nd line treatments. Deep discounts are required for all new agents in order to achieve cost-effectiveness thresholds between $100,000 and $150,000 per QALY gained. As noted previously, the budgetary impact of each of these regimens does not exceed our stated threshold when annualized over a five-year time horizon. Therefore, the draft ICER value-based price benchmark for 2nd line CFZ, with all the assumptions mentioned previously regarding net costs, is $673 to $1,267 per vial. This price represents a 32%-64% discount from the estimated cost of CFZ. The draft ICER value-based price benchmark for 2nd line ELO is $267 to $588 per 400 mg vial, representing a 75%-89% discount from the cost per vial. The draft ICER value-based price benchmark for 2nd line IX is $181 to $587 per capsule, representing an 80%-94% discount from the WAC price.
Draft Value-based Price Benchmarks for Second-Line Regimens
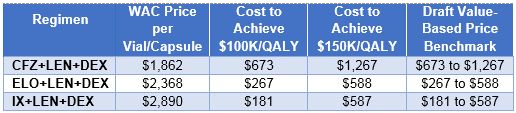
Table for third- line treatments is available in the report on page 61.
The introduction of newer regimens for second- and third-line use in multiple myeloma appears to confer clinical benefits in terms of lengthening progression-free and overall survival as well as improved quality of life. However, at current wholesale acquisition costs, the estimated cost-effectiveness of these regimens exceeds commonly-cited thresholds. Achieving levels of value more closely aligned with patient benefit would require substantial discounts from the list price in many cases, and in other cases, there is no realistic price for the newest agents that would achieve these thresholds.
